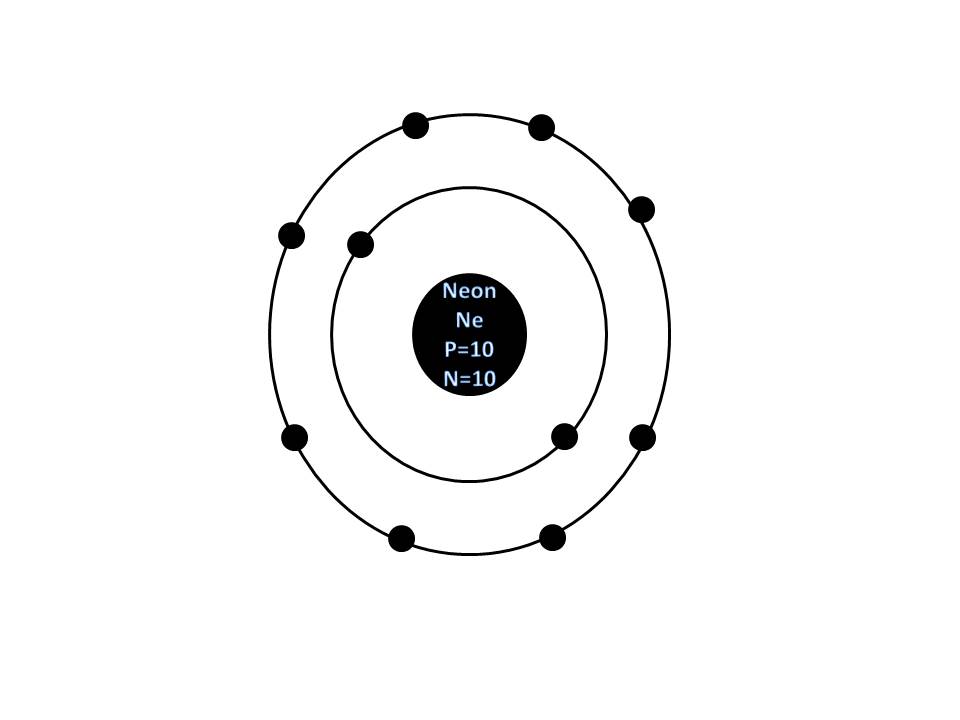
The ground state energy of the electrons 1s 2. Under this condition of ionizations I suggest that n = 1īecomes n 0.9994, because the second electron increase the electronĭetailed knowledge about the electromagnetic force between the two spinningĮlectrons of opposite spin physicist today using wrong theories cannot explain One sees here that after the ionizations the Bohr model gives the value ofġ360.57 eV which is smaller than the experimental value of E 10 =ġ362.1995. Thus we expect to calculate it byĪpplying the simple Bohr model for Z = 10 as In the case of the helium the ionization energy E 10 =ġ362.1995 eV = - E(1s 1) is due to the one remainingĮlectron of 1s 1 with n = 1. Of the 1s 2 electrons one must apply both the Bohr model and my formulaĮXPLANATION OF Ε 10 = 1362.1995 eV AND E 9 = 1195.8286 eV, WHICH GIVE THE GROUND STATE ENERGY OF 1s 2 ELECTRONS Of the E 9 and E 10 which give the ground state energy Ground state energies of (2s 2.2p x 2.2p y 2.2p z 2) you can see my “ EXPLANATION OF NEON IONIZATIONS”. Interactions of electrons and also of nucleons create atomic molecular and See also my papers about the explanation of ionization energies of theĮlements in my FUNDAMENTAL PHYSICS CONCEPTS based on my paper “ Spin-spin 
The ionization energies (in eV) of the neon atom are the following: E 1 =ġ362.1995. Under this condition the correct electron configuration should be give by this image of Neon including the following electron configuration: 1s 2.2s 2.2p x 2.2p y 2.2p z 2.Īccording to the “ Ionization energies of the elements-WIKIPEDIA” Of two spinning electrons of opposite spin. Interactions of electrons and also of nucleons create atomic molecular and nuclear structures” (2008) by analyzing carefully the electromagnetic interactions Under these difficulties I published my paper “ Spin-spin The Hartree–Fock method, could be used to estimate the ground state energies. However unlike for hydrogen, aĬlosed-form solution to the Schrödinger equation for the many-electron atoms Neon is an atom of the chemical element neon with symbol Ne and atomic number 10. Kaliambos ( Natural Philosopher in New Energy) The electronic configuration of Neon will be 1s2 2s2 2p6.By Prof. How do you write the electron configuration for Neon? The electronic configuration of Neon will be 1s2 2s2 2p6. What is the electronic configuration of Neon 10? What is the boiling Point of Neon in Kelvin?īoiling Point of Neon in Kelvin is 27.07 K. Melting Point of Neon in Kelvin is 24.56 K. What is the melting Point of Neon in Kelvin? What is the boiling Point of Neon?īoiling Point of Neon is 27.07 K. Neon has 10 electrons out of which 0 valence electrons are present in the 2s2 2p6 outer orbitals of atom. How many valence electrons does a Neon atom have? It is located in group 18 and period 2 in the modern periodic table. Neon is the 10 element on the periodic table. Neon is a chemical element with the symbol Ne and atomic number 10. What is the position of Neon in the Periodic Table? Neon is a chemical element with symbol Ne and atomic number 10. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Neon is 2s2 2p6. What is the abbreviated electronic configuration of Neon? 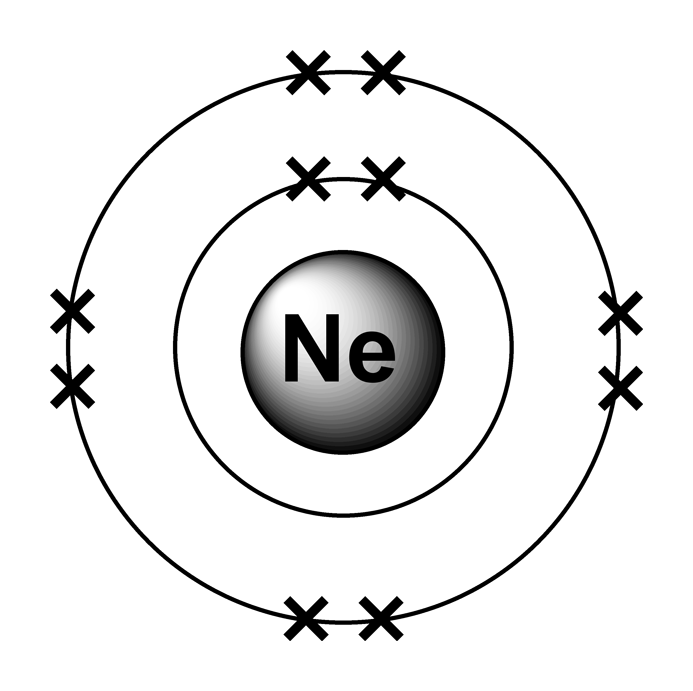
The electronic configuration of Neon is 1s2 2s2 2p6. What is the electronic configuration of Neon?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
